 High Precision Auto Diagnostic Tool Garage Equipment Key Programmer
High Precision Auto Diagnostic Tool Garage Equipment Key Programmer
 Km-C01 Smoke Leak Detector Tester Car Diagnostic Tool
Km-C01 Smoke Leak Detector Tester Car Diagnostic Tool
 Smoke Detector Leak Car Km-01 Diagnostic Machine for Evap System
Smoke Detector Leak Car Km-01 Diagnostic Machine for Evap System
 Launch X-431 Adas Mobile Portable Automotive Diagnostic Tools
Launch X-431 Adas Mobile Portable Automotive Diagnostic Tools
 Suzuki 4-Pin Diagnostic Harness for Motorcycle ECU Reading
Suzuki 4-Pin Diagnostic Harness for Motorcycle ECU Reading
 Clinical Urine Analyzer Automated System with 11-Parameter Testing
Clinical Urine Analyzer Automated System with 11-Parameter Testing
 Digital EEG Brain Mapping System Medical Diagnostic Equipment
Digital EEG Brain Mapping System Medical Diagnostic Equipment
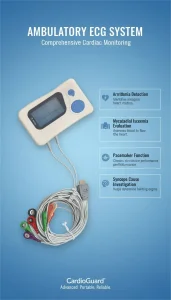 Portable Medical Dynamic ECG Monitoring System with Holter Analysis
Portable Medical Dynamic ECG Monitoring System with Holter Analysis
The global market for handheld diagnostic devices has entered a transformative era. Driven by the urgent need for rapid, on-site data analysis, industries ranging from clinical healthcare to automotive engineering are moving away from centralized lab testing toward Point-of-Care Testing (POCT) and mobile field diagnostics. As leading OEM/ODM Handheld Diagnostic Devices Suppliers & Exporters, we have observed a 40% increase in demand for portable, high-precision tools over the last three years.
In the commercial sector, the rise of electric vehicles (EVs) and complex medical imaging has necessitated devices that are not only portable but also capable of processing massive amounts of telemetry and biological data. The shift is clear: efficiency is no longer optional; it is the core of industrial competition.
Modern diagnostic tools are no longer passive readers. Integration of AI allows for predictive maintenance in automotive scanners and automated morphology analysis in medical hematology systems, reducing human error by up to 95%.
Real-time data synchronization via 5G and Wi-Fi 6 enables technicians and clinicians to share diagnostic reports instantly with cloud servers, facilitating remote consultation and fleet management.
There is a growing push for "Green Diagnostics." Our manufacturing process focuses on low-power consumption hardware and recyclable high-precision acrylic components to meet global ESG standards.
For global procurement officers and distributors, selecting a reliable ODM diagnostic device partner involves more than just comparing price tags. Reliable procurement focuses on three pillars:
Our handheld devices are engineered for diverse environments. In South East Asia, our infectious disease diagnostic kits are used in remote clinics with limited power. In Europe and North America, our high-precision automotive tools like the Launch X-431 series are the backbone of professional independent workshops and ADAS calibration centers.
As a premier high-tech biotechnology company based in Ningbo, China, Ningbo Medvok Medical Co., Ltd. offers unparalleled advantages in the handheld diagnostic sector. China's mature industrial ecosystem allows us to integrate sophisticated electronics with advanced biological raw materials at a speed and cost-efficiency unmatched in the West.
Ningbo Medvok Medical Co., Ltd. is a high-tech biotechnology company focused on the R&D and production of antigens, antibodies and downstream detection reagents for diagnosis and therapy. The product pipelines cover cardiovascular and cerebrovascular, inflammation, infectious diseases, tumors, hormones and other categories, from raw materials to finished products.

Innovation is in our DNA! Bioantibody keeps developing new technologies. Currently, our products have been delivered to more than 60 countries and cities worldwide. Using the ISO 13485 management system, the product quality is greatly trusted by customers. With the mission [Biotech For A Better Life], we are committed to innovation and providing our best solutions to our customers. We sincerely believe we could make our special contribution to human ecology and health.
By controlling the entire value chain—from the molecular level of antigens to the final handheld casing—we ensure 100% quality traceability. This vertical integration is why we are the preferred OEM/ODM Handheld Diagnostic Devices Exporter for clinical and industrial sectors alike.

Our authority in the diagnostic field is backed by years of intensive research. As an IVD solutions provider, we have worked and will keep working intensively to support research and foment knowledge and prevention of the diseases. For us, a well-informed society is a healthier society.
We care about protecting the human health and hope all of society to have access to clean, affordable, and reliable biotechnologies. Furthermore, our economic development will be compatible with proper conduct in relation to ethics, society, the workplace, the environment and respect for human rights. We think of society as a group of individuals with equal rights and opportunities.
In order to materialize this commitment, we have developed the sustainability policy on environmental and social matters, ensuring that every diagnostic tool exported from our facility contributes to a better world.
 Automated Digital Hematology Diagnostic Equipment
Automated Digital Hematology Diagnostic Equipment
 Precision Acrylic and Plastic Manufacturing for Analyzers
Precision Acrylic and Plastic Manufacturing for Analyzers
 Launch X-431 Pad VII Automotive Diagnostic Tool
Launch X-431 Pad VII Automotive Diagnostic Tool
 High Precision Automotive Diagnostic Tool with 10.1 Screen
High Precision Automotive Diagnostic Tool with 10.1 Screen
 High Precision Car Diagnostic Tools for Auto Repair
High Precision Car Diagnostic Tools for Auto Repair
 Sterilizing Pharmaceutical Diagnostic Reagent Preparation
Sterilizing Pharmaceutical Diagnostic Reagent Preparation
 BPE Standard Easy to Clean Reagent Preparation System
BPE Standard Easy to Clean Reagent Preparation System
 No Sediment High Efficiency Reagent Preparation System
No Sediment High Efficiency Reagent Preparation System