
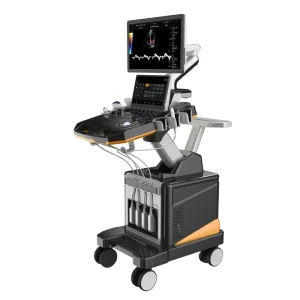
Umus021 Dual-Screen Color Ultrasound System - High-Resolution Imaging

Smart Greenhouse Solutions for Energy Efficiency and Sustainability

Tailored Automation Solutions to Boost Performance and Reliability

Smart Health Monitoring Sensors for Customized Medical Solutions

ASA Plastic Moulding Solutions for Industrial Snap Fasteners


Advanced CNC Machining Solutions with Smart Automation Technology
In the rapidly evolving world of modern healthcare, Point-of-Care (POC) Connectivity Solutions have transitioned from a luxury to a critical necessity. As the global healthcare market moves toward decentralized testing, the ability to connect diagnostic devices directly to Hospital Information Systems (HIS) and Electronic Medical Records (EMR) is paramount.
Today, medical facilities from New York to Shanghai are demanding real-time data synchronization. The global POC market is projected to reach unprecedented heights, driven by the need for rapid diagnosis in emergency rooms, remote monitoring in elderly care, and precise data management in surgical suites. Our role as a leading OEM supplier is to bridge the gap between sophisticated bio-hardware and seamless digital ecosystems.
From the early days of manual data entry to the current era of automated IoT integration, POC connectivity has revolutionized how clinical decisions are made. By reducing human error and accelerating turnaround times, these solutions are the backbone of the "Smart Hospital" initiative worldwide.
The integration of 5G technology allows for near-zero latency in data transmission, enabling remote robotic surgeries and real-time ultrasound streaming over vast distances.
Connectivity isn't just about moving data; it's about processing it. Our OEM solutions incorporate AI middleware to flag abnormal results before they even reach the doctor's screen.
With data breaches on the rise, robust encryption and compliance with international standards like GDPR and HIPAA are at the core of our connectivity architecture.
Standardization via HL7, FHIR, and DICOM ensures that our POC devices communicate effortlessly with any existing medical infrastructure globally.
Global procurement officers are no longer looking for standalone devices; they are seeking integrated ecosystems. Whether it is a government-funded rural health initiative in Southeast Asia or a private hospital group in Europe, the requirements are consistent: scalability, reliability, and cost-effectiveness.
Ningbo Medvok Medical Co., Ltd. is a high-tech biotechnology company focused on the R&D and production of antigens, antibodies and downstream detection reagents for diagnosis and therapy. The product pipelines cover cardiovascular and cerebrovascular, inflammation, infectious diseases, tumors, hormones and other categories, from raw materials to finished products.

Innovation is in our DNA! Bioantibody keeps developing new technologies. Currently, our products have been delivered to more than 60 countries and cities worldwide. Using the ISO 13485 management system, the product quality is greatly trusted by customers. With the mission "Biotech For A Better Life", we are committed to innovation and providing our best solutions to our customers. We sincerely believe we could make our special contribution to human ecology and health.

As an IVD solutions provider, we have worked and will keep working intensively to support research and foment knowledge and prevention of the diseases. For us, a well-informed society is a healthier society. We care about protecting the human health and hope all of society to have access to clean, affordable, and reliable biotechnologies.
Furthermore, our economic development will be compatible with proper conduct in relation to ethics, society, the workplace, the environment and respect for human rights. We think of society as a group of individuals with equal rights and opportunities. In order to materialize this commitment, we have developed the sustainability policy on environmental and social matters.
In the ICU, every second counts. Our POC connectivity solutions allow ventilators, infusion pumps, and patient monitors to stream data to a central nursing station, triggering automated alerts if patient vitals deviate from the norm.
Equipping ambulances with connected ultrasound and blood gas analyzers means hospital specialists can view diagnostic images while the patient is still in transit, preparing the OR before the ambulance even arrives.
For regions with limited specialist access, our cloud-connected tablets and sensors allow local practitioners to send diagnostic data to metropolitan experts for instant consultation, bridging the healthcare divide.
Beyond the bedside, our connectivity frameworks streamline IVD reagent tracking and result validation, ensuring that every laboratory test is logged with a complete digital audit trail.
With years of experience in both biological raw materials and precision medical hardware, we offer a "vertical" OEM service that few can match—controlling quality from the molecular level to the final digital handshake.
Our commitment to E-E-A-T (Experience, Expertise, Authoritativeness, and Trustworthiness) ensures that every project we undertake is backed by scientific rigor and industrial excellence. From high-resolution ultrasound systems to smart health monitoring sensors, we deliver the future of medical connectivity today.