














| Model NO. | Diagnostic reagent preparation system |
| Power Source | Electric |
| Safety Features | Emergency Stop Button, Overload Protection, Safety Interlock |
| Warranty | 1 Year |
| Precision | High Precision |
| Computerized | Computerized |
| Condition | New |
| Monitoring System | Equipped |
| Transport Package | Exporting Wooden Package |
| Specification | Customized |
| Origin | China |
| Production Capacity | 2000 sets Per Year |

High-Capacity Automatic Sterile Diagnostic reagent preparation system
This system is specially designed for diagnostic reagent preparation.






Closed tanks, aseptic connectors, and disposable technologies avoid environmental exposure. Meets EU GMP Annex 1. Supports Online Sterilization (SIP) and integration of 0.22μm filters.
Dual control of weighing and flow via mass flowmeters. Real-time online monitoring of pH, conductivity, and temperature. Advanced formula management software with hierarchical permissions.
Material parts are 316L stainless steel or USP Class VI disposable materials. Complies with FDA 21 CFR Part 11 and ALCOA+ principles for data integrity.
PLC/SCADA control for automated execution of formula steps. IoT-based remote monitoring and predictive maintenance. Automatic electronic record generation.

| Application | Pharmaceutical, Biotechnology, Healthcare |
| Capacity | Customized |
| Process Type | Liquid preparation system |
| Specification | Customized |



A leading pharmaceutical equipment manufacturer, specializing in energy-efficient and high-performance pharmaceutical water and liquid preparation system solutions. Widely used in biopharmaceuticals, medical devices, medical aesthetics, food and beverage, and electronics industries.
Committed to delivering solutions that comply with FDA, cGMP, and QS standards. Products include pure water systems, water for injection systems, aseptic preparation systems, and oral liquid systems. Full validation documentation sets (URS, FAT, SAT, DQ, OQ, IQ, PQ) are provided.
ISO9001 and CE certifications, pressure-bearing equipment installation qualifications. Production complies with high-standard GMP requirements.
316L stainless steel construction with automated control to improve production efficiency and ensure corrosion resistance.
24-hour rapid response, regular maintenance plans, and technical training. Support for GMP certification and document preparation.
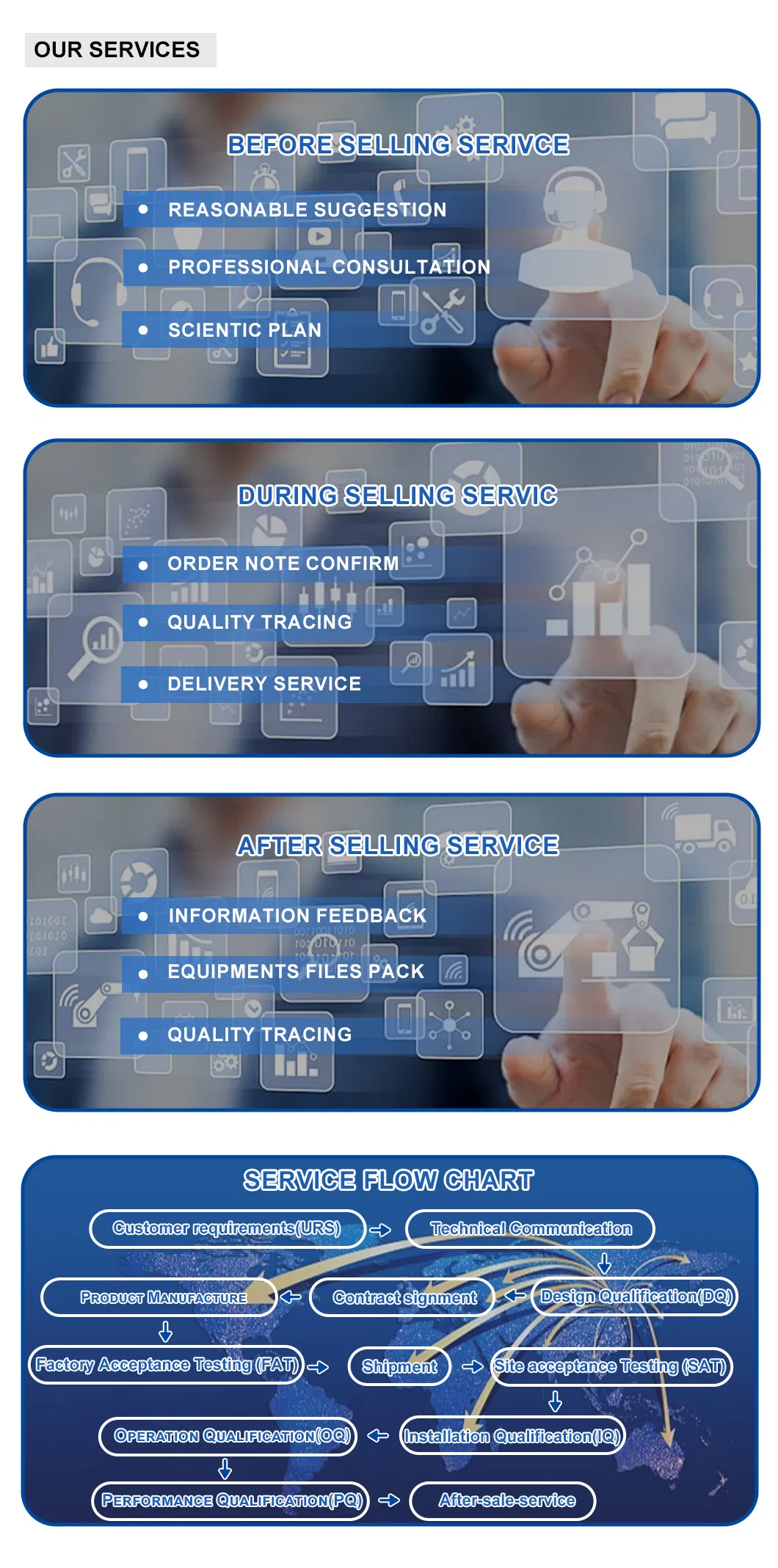



Yes, all equipment is designed and manufactured in accordance with GMP, FDA, and WHO requirements specific to your region.
Standard products typically take 4-6 weeks. For customized systems, the lead time is 6-12 weeks depending on the technical complexity.
We offer a 12-month warranty against manufacturing defects. Extended warranties and comprehensive maintenance plans are also available.
Our 24/7 service team provides support via email, phone, or video call. Global spare parts shipping is available within 3-7 days.
We provide remote troubleshooting first. If required, specialized engineers can be deployed for on-site visits to resolve complex issues.
Yes, we provide a full set of validation documents including URS, FAT, SAT, DQ, OQ, IQ, and PQ to help you meet regulatory requirements.