This system is specially designed for diagnostic reagent preparation, featuring customer-customized three-layer insulation for optimal performance.






Closed tanks, aseptic connectors, and disposable technologies reduce contamination risk. Includes Online Sterilization (SIP) and 0.22μm aseptic filtration integration.
Dual control of weighing and flow via high-precision modules and mass flowmeters. Real-time online monitoring of pH, conductivity, and temperature.
Complies with FDA 21 CFR Part 11 and ALCOA+ principles. 316L stainless steel materials meet USP Class VI requirements.
PLC/SCADA control for automated execution of formula steps. Features remote monitoring, predictive maintenance, and automatic record generation.


| Application | Pharmaceutical, Biotechnology, Healthcare |
| Capacity | Customized based on client requirements |
| Process Type | Liquid preparation system |
| Main Materials | 316L Stainless Steel / USP Class VI Disposable materials |
| Control System | PLC / SCADA with Audit Trail |
We are a leading pharmaceutical equipment provider, specializing in energy-efficient pharmaceutical water and liquid preparation systems. Our solutions comply with FDA, cGMP, and QS standards, serving biopharmaceuticals, medical aesthetics, and electronics industries worldwide.

High-tech enterprise with ISO9001 and CE certifications. All equipment complies with GMP standards for high-safety industries.
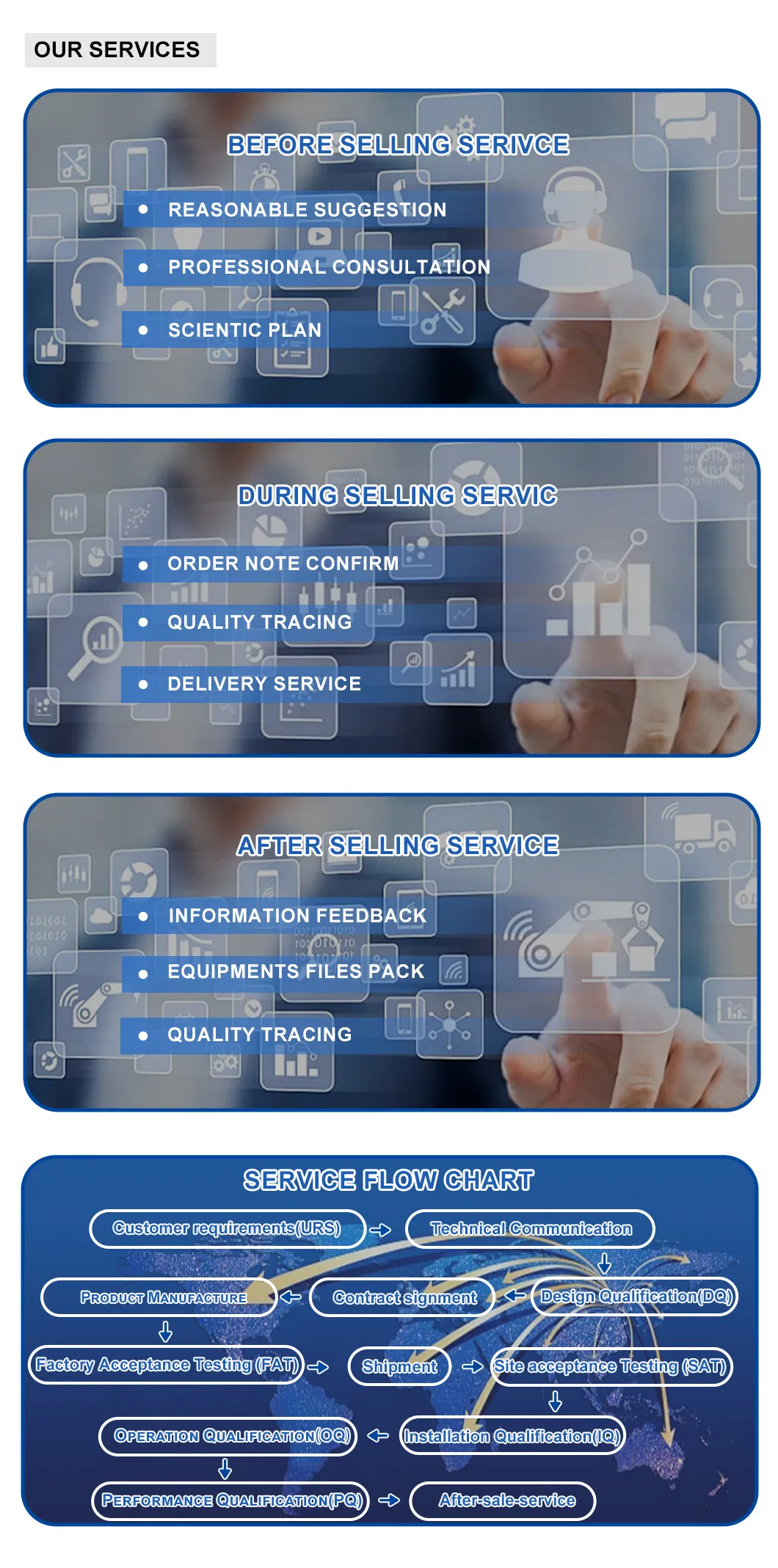
24-hour rapid response, regular maintenance plans, and technical training. Support for full-process validation (URS, FAT, SAT, DQ, IQ, OQ, PQ).