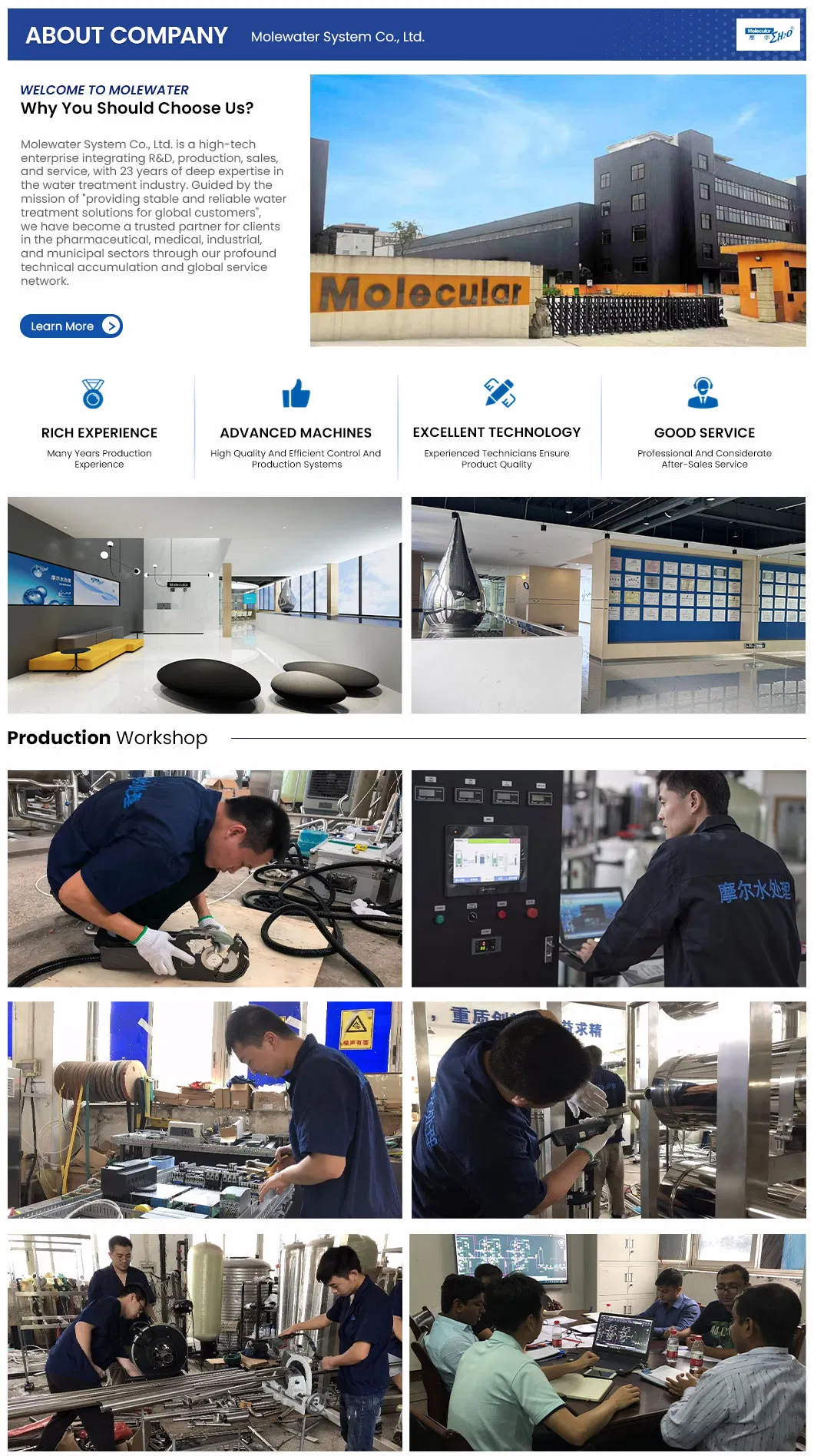
Pharmaceutical Purified Water System, WFI Multi-effect Distiller, and Pure Steam Generator.
We provide comprehensive turn-key solutions for pharmaceutical water system projects, covering technical design, high-precision manufacturing, global shipping, on-site installation, and professional operational training.
| No. | Unit | Parameter Information |
|---|---|---|
| 1 | Name | 3000L/H Purified Water (PW) System |
| 2 | Model | MOLPW-3000L |
| 3 | Permeate Capacity | 3000L/H @ 25℃ |
| 4 | Feed Water | Municipal Tap Water |
| 5 | Pure Water Quality | Electrical Conductivity < 1.3μs/cm (Meets GMP, EP/USP) |
| 6 | Technology | Double Stage Reverse Osmosis + EDI Unit |
| 7 | Control Method | Automatic PLC Control (Manual override available) |

Our systems integrate cutting-edge Reverse Osmosis (RO) and Electrodeionization (EDI) technology to consistently produce ultra-pure water that exceeds global pharmacopoeia requirements with a significant safety margin.
Equipped with a high-end PLC+HMI control system, the equipment offers real-time monitoring and intelligent algorithms to optimize operational parameters, maximizing efficiency while minimizing manual intervention.


The modular construction simplifies installation and future expansion. All contact components are pharmaceutical-grade stainless steel with a dead-leg-free design to prevent microbial growth.
Features include advanced alarm systems, self-diagnostics, pressure relief valves, and abnormal water quality alerts to ensure continuous safe operation for critical processes.


Smart optimization reduces energy consumption by up to 30% compared to conventional systems. The EDI module significantly lowers operational costs and environmental impact.